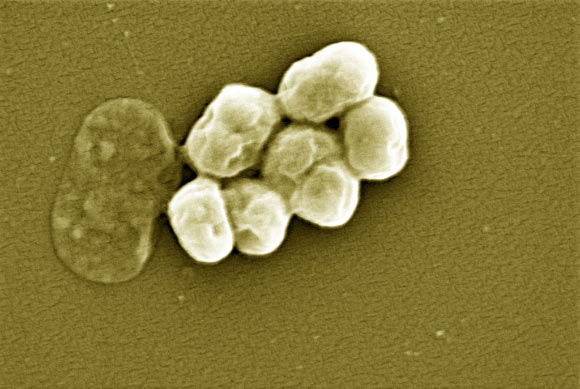
Nourseothricin, also known as the streptothricin natural product mixture, was discovered in the early 1940s, generating intense initial interest because of excellent activity against Gram-negative bacteria.
Nourseothricin is a natural product made by Streptomyces soil fungi, which contains multiple forms of a complex molecule called streptothricin.
Its discovery in the 1940s generated high hopes for it as a powerful agent against Gram-negative bacteria, which, due to their thick outer protective layer, are especially hard to kill with other antibiotics.
But nourseothricin proved toxic to kidneys, and its development was dropped.
However, the rise of antibiotic-resistant bacterial infections has spurred the search for new antibiotics, leading Dr. James Kirby, a researcher at Beth Israel Deaconess Medical Center and Harvard Medical School, and his colleagues to take another look at nourseothricin.
Early studies of nourseothricin suffered from incomplete purification of the streptothricins.
More recent work has shown that the multiple forms have different toxicities with one, streptothricin-F, significantly less toxic, while remaining highly active against contemporary multidrug-resistant pathogens.
In the new study, the authors characterized the antibacterial action, renal toxicity, and mechanism of action of highly purified forms of two different streptothricins, D and F.
The D form was more powerful than the F form against drug-resistant Enterobacterales and other bacterial species, but caused renal toxicity at a lower dose. Both were highly selective for Gram-negative bacteria.
Using cryo-electron microscopy, the researchers showed that streptothricin-F bound extensively to a subunit of the bacterial ribosome, accounting for the translation errors these antibiotics are known to induce in their target bacteria.
Interestingly, the binding interaction is distinct from other known inhibitors of translation, suggesting it may find use when those agents are not…
Read the full article here