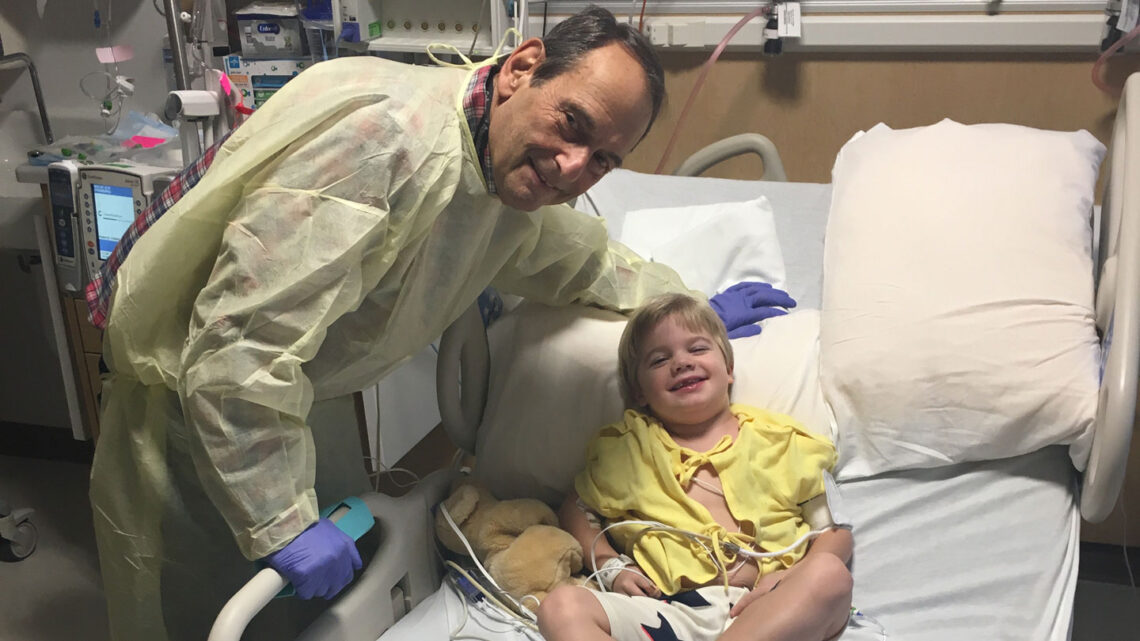
Will Ungerer isn’t a typical fourth-grader.
The 10-year-old from Midlothian, Va., plays tag with his friends and swims in the ocean. “I think his peers would describe him as someone who is kind, sweet and funny, and supersmart,” says his mother, Sheila Ungerer. He won a countywide citizenship award and was named classroom student of the year.
But “he’s not a really fast runner,” his peers might say. “He doesn’t really run.” Will was born with Duchenne muscular dystrophy, a genetic disease that causes muscles throughout the body to break down over time. That includes not only skeletal muscles, but also the heart and diaphragm, which controls breathing. Eventually those important muscles stop working properly, leading to death. The disease is caused by mutations in the gene responsible for dystrophin, a protein that acts as a shock absorber to prevent damage as muscles contract. It affects an estimated 4.8 out of 100,000 people worldwide, mainly boys and young men. About 10,000 people in the United States have Duchenne muscular dystrophy.
Will celebrated his birthday last year at a laser tag park, his mother says, but other kids his age with Duchenne’s “don’t ride the bus. They don’t carry their tray in the lunchroom.”
The difference is that Will received an experimental gene therapy when he was 5 years old. Just before Christmas in 2018, doctors at Nationwide Children’s Hospital in Columbus, Ohio, gave Will an infusion of viruses. Those viruses delivered instructions to his muscles for making a short form of the dystrophin protein. Scientists at Sarepta Therapeutics in Cambridge, Mass., the company that makes the gene therapy, developed this version, called microdystrophin, to act as a replacement, hopefully protecting muscles from harm.
Though the therapy did not show statistically meaningful improvements compared with a placebo in one randomized controlled trial and results of an ongoing clinical trial to…
Read the full article here