The carbon atom provides the backbone for the complex organic chemistry composing the building blocks of life. The physics of the carbon nucleus in its predominant isotope, carbon-12, is similarly full of multifaceted complexity. In new research, physicists from Michigan State University and elsewhere examined the structure of the low-energy nuclear states of carbon-12 using nuclear lattice effective field theory.
The element carbon is critical to organic chemistry and life as we know it. The physics of its most common isotope, carbon-12, is extremely complex.
Many experimental and theoretical investigations have been devoted to determining the energies and underlying structures of the nuclear states of carbon-12.
In the new study, Michigan State University physicist Dean Lee and colleagues computed these states from first principles — the most basic components of physics theory.
They used supercomputers and nuclear lattice simulations to calculate the three-dimensional shape formed by the protons and neutrons comprising the nucleus.
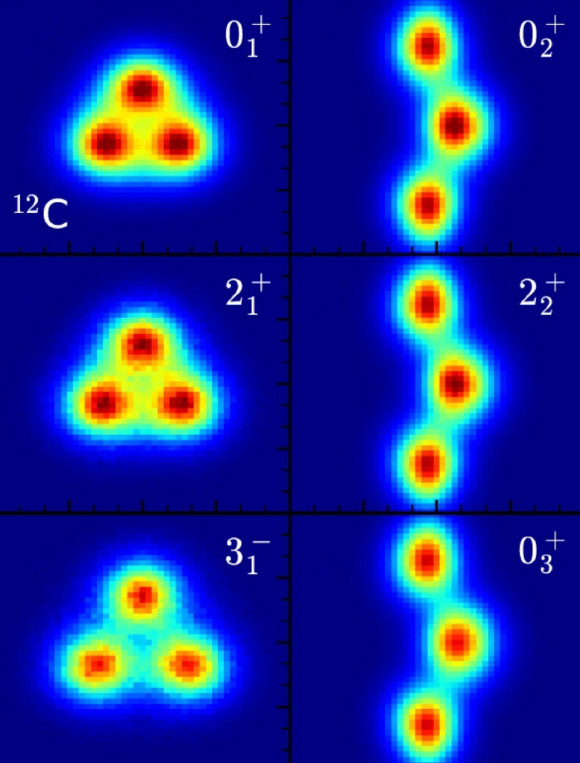
Their results show that all of the low-lying energy states of carbon-12 have a substructure where the six protons and six neutrons cluster together into alpha particles.
Alpha particles are helium-4 nuclei, which contain two protons and two neutrons.
“One well-known nuclear state of carbon-12 is the Hoyle state,” the physicists said.
“This state has an energy that sits near the energy threshold for three alpha particles or helium nuclei.”
“This energy thereby greatly enhances the production of carbon in helium-burning stars. This helps to explain the presence of carbon in the Universe.”
“Our results show that the Hoyle state is composed of a ‘bent arm’ or obtuse triangular arrangement of alpha particles.”
“All the low-lying energy states of carbon-12 have an intrinsic shape composed of three alpha particles forming either an equilateral triangle or an obtuse triangle.”
The team’s results give…
Read the full article here