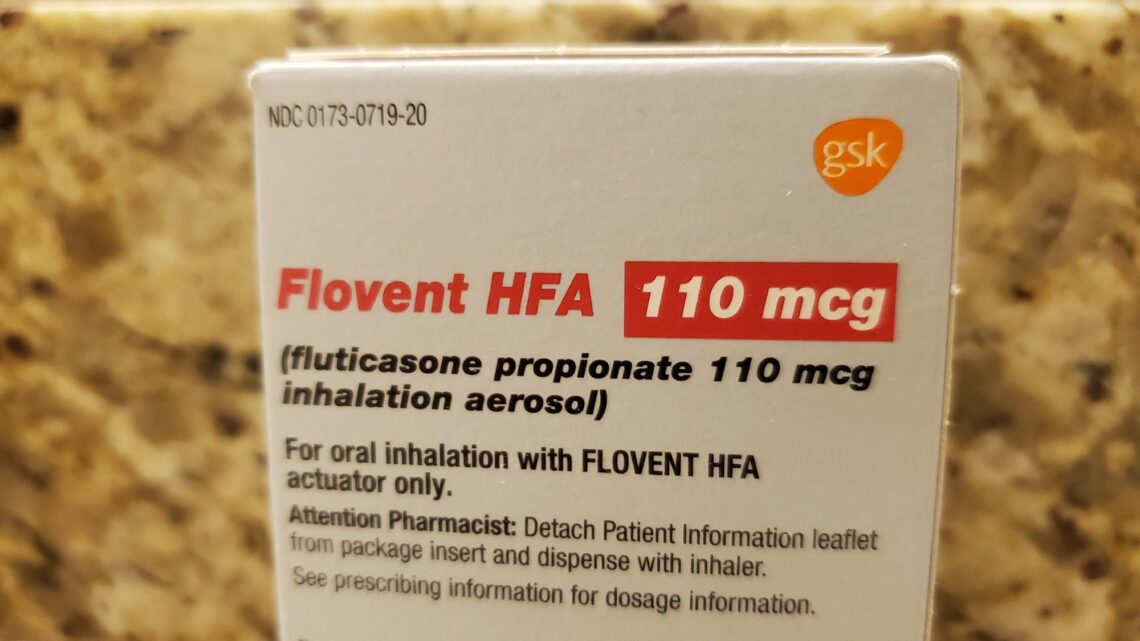
On January 1, pharmaceutical company GlaxoSmithKline (GSK) discontinued Flovent, a popular steroid inhaler that is used to lower the frequency of asthma attacks and other symptoms. The company discontinued production of the branded product in preparation for the rollout of an “authorized generic” version of the widely prescribed medication. The Asthma and Allergy Foundation of America estimates that about 25 million people have asthma in the United States and using inhaled corticosteroids like Flovent are among the most effective treatments for the disease. Here’s what you should know.
[Related: Gas stoves could be making thousands of children in America sick.]
What is Flovent?
Flovent is the brand name of fluticasone, a medication that has been sold since 2020. It is an inhaled corticosteroid that can be used by people four years of age and older for treating asthma long term. Fluticasone helps to keep inflammation in the lungs low and the airways open. It is used twice per day and is not a rescue inhaler. When it is taken every day, it can lower the number and severity of asthma attacks.
GSK first notified the FDA about the decision to stop manufacturing Flovent in June 2023.
Why is there a new ‘authorized generic’ version?
According to a statement from GSK to the Allergy Foundation of America, the generic version of the prescription inhaler will have an identical formula and drug-delivery mechanism. GSK says that this new version will provide lower cost alternatives.
However, this switch from the branded version to a generic one occurs at the same time as the elimination of the rebate cap removal of Medicaid drug prices. This provision was made as part of the American Rescue Plan Act of 2021. Under it, pharmaceutical companies are required to pay states higher Medicaid rebates if they raise the cost of the drug more than inflation. According to GoodRx, Flovent’s average price increased from about $207 in 2013…
Read the full article here