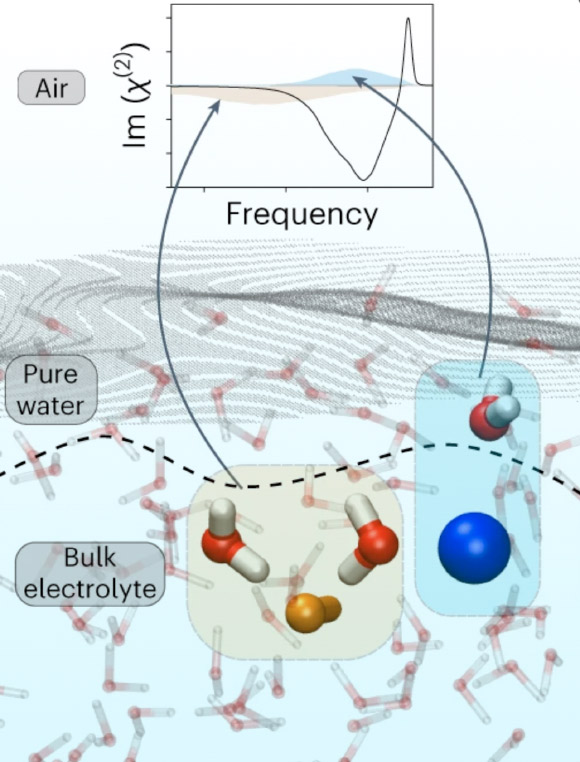
The distribution of ions at the air/water interface plays a decisive role in many natural processes. Several studies have reported that larger ions tend to be surface-active, implying ions are located on top of the water surface, thereby inducing electric fields that determine the interfacial water structure. However, new research by chemists from the University of Cambridge and the Max Planck Institute for Polymer Research challenges this view. Their results show that ions in typical electrolyte solutions are, in fact, located in a subsurface region, leading to a stratification of such interfaces into two distinctive water layers.
Many important reactions related to climate and environmental processes take place where water molecules interface with air.
For example, the evaporation of ocean water plays an important role in atmospheric chemistry and climate science.
Understanding these reactions is crucial to efforts to mitigate the human effect on our planet.
The distribution of ions at the interface of air and water can affect atmospheric processes. However, a precise understanding of the microscopic reactions at these important interfaces has so far been intensely debated.
University of Cambridge’s Dr. Yair Litman and colleagues set out to study how water molecules are affected by the distribution of ions at the exact point where air and water meet.
Traditionally, this has been done with a technique called vibrational sum-frequency generation (VSFG).
With this laser radiation technique, it is possible to measure molecular vibrations directly at these key interfaces.
However, although the strength of the signals can be measured, the technique does not measure whether the signals are positive or negative, which has made it difficult to interpret findings in the past. Additionally, using experimental data alone can give ambiguous results.
The authors overcame these challenges by utilizing a more sophisticated form of VSFG, called heterodyne-detected…
Read the full article here