This article was originally featured on Knowable Magazine.
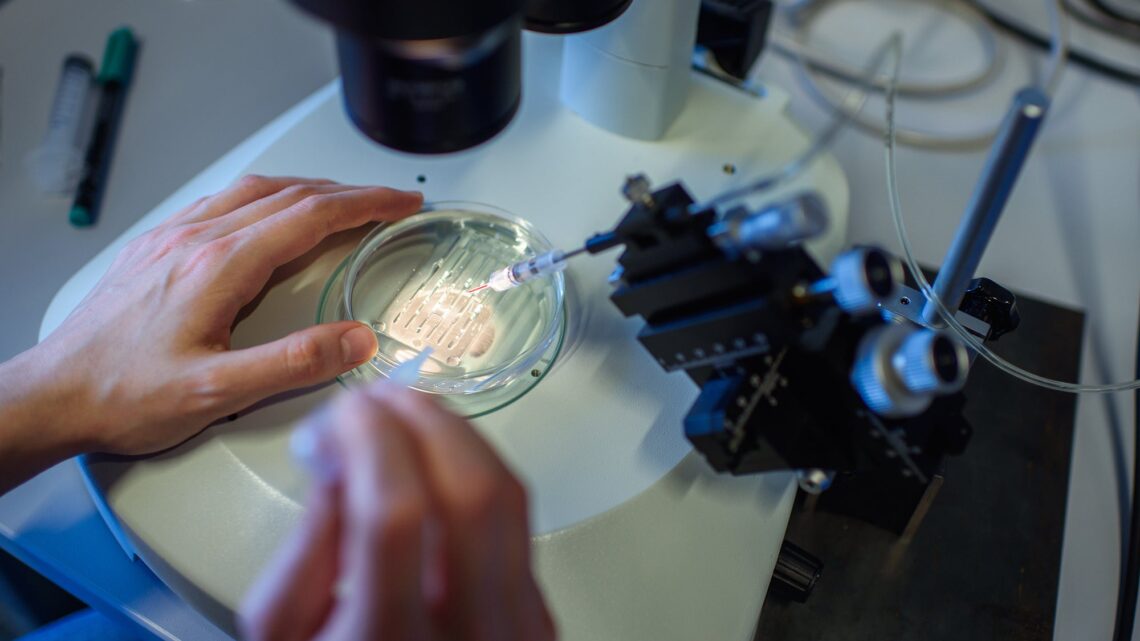
Around the turn of the century, microbiologists at Danisco USA Inc. had a problem: The bacteria they used to make yogurt and cheese were getting infected with viruses. Investigating more deeply, the scientists found that some bacteria possessed a defense system to fight off such viral invaders.
These virus-resistant bacteria carried weird, repetitive collections of DNA letters in their chromosomes—bits of DNA from their encounters with past viruses that the microbes had “saved” in their own genomes. It was a form of molecular memory akin to the way that our own immune system remembers invaders so it can make antibodies against a recurring infection.
In this case, the microbes’ immune system, dubbed CRISPR-Cas, or more casually, just CRISPR, shreds any viral genome that matches the sequences in their molecular memory banks.
The yogurt-makers weren’t looking for biotechnology’s Next Big Tool. They just wanted to preserve the products in their vats. But other scientists soon realized the potential value of CRISPR for their own designs: With some modifications, CRISPR allowed them to cut any genetic sequence they wanted to, greatly easing the challenge of genetic engineering.
CRISPR systems have taken the biotechnology world by storm, nabbing a Nobel Prize and kicking off a new era of gene therapy. In December 2023, the US Food and Drug Administration approved the first CRISPR-based gene-changing treatment: a new gene therapy for the excruciatingly painful blood disorder sickle cell anemia.
“It’s been revolutionary for research,” says Kevin Esvelt, an evolutionary engineer at the MIT Media Lab in Cambridge, Massachusetts. “It’s accelerating all of biotech.”
Read the full article here