- For the first time, a highly effective new malaria vaccine — RTS,S/AS01 — has rolled out in Cameroon, Africa.
- RTS,S/AS01 has shown modest efficacy against symptomatic malaria in clinical trials.
- The GSK-developed vaccine, also known as Mosquirix, is one of two malaria vaccines to receive approval from the World Health Organization (WHO).
- WHO has also recommended the R21/Matrix-M vaccine, which shows 75% effectiveness against symptomatic malaria over 24 months.
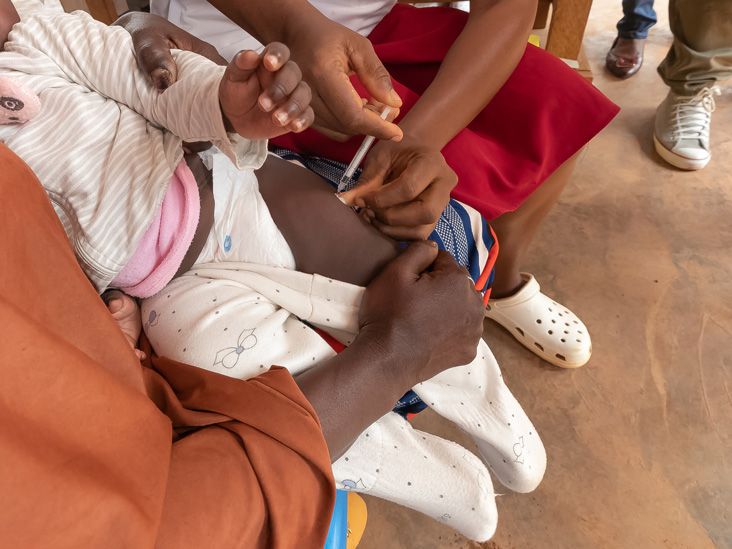
An effective new malaria vaccine was rolled out for the first time in Cameroon, Africa, on January 22.
Global health officials say the immunization campaign for the RTS,S vaccine (Mosquirix) marks a historic moment in the fight against this mosquito-borne disease, one of the most life threatening infections in the world.
The vaccine drive in Cameroon is the first official public launch of the Mosquirix vaccine outside of clinical trials, which have shown a 13% reduction in all-cause mortality, including malaria-related deaths, in children who were eligible to receive it.
At least 20 African countries have made plans for malaria vaccine rollouts as cases and deaths related to the disease continue to climb. The majority of malaria cases occur on the continent, with an estimated
According to the Africa CDC, in 2016, there were around 216 million cases of malaria in 91 countries globally and 445,000 deaths. That same year, 90% of malaria cases and 91% of malaria deaths occurred in the WHO AFRO Region.
But malaria is both preventable and curable, especially with the help of the new vaccine.
“We are not just witnessing, but actively participating in a transformative chapter in African public health history,” said Mohammed Abdulaziz, the head of disease control and prevention at Africa CDC, at a recent press conference.
“For a long time, we have been waiting for a day like this. It brings more than just hope. It brings a reduction in the mortality and morbidity…
Read the full article here