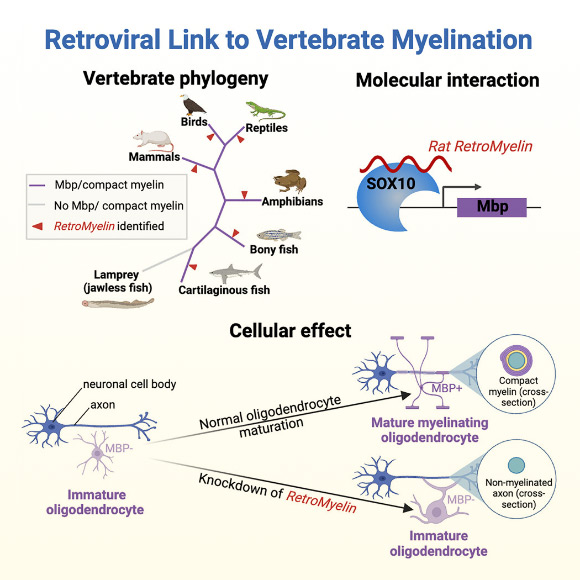
Biologists from Altos Labs-Cambridge Institute of Science, the University of Cambridge and elsewhere have found that a retrovirus-derived genetic element (retrotransposon) is essential for the production of myelin — the insulating sheath that surrounds neuronal axons — in mammals, amphibians, and fish. Dubbed RetroMyelin, this gene sequence is likely a result of ancient retroviral infection, and comparisons of RetroMyelin in mammals, amphibians, and fish suggest that retroviral infections and genome-invasion events occurred separately in each of these groups.
Myelin — a complex, fatty tissue that ensheathes vertebrate nerve axons — enables rapid impulse conduction without needing to increase axonal diameter, which means nerves can be packed closer together.
It also provides metabolic support to nerves, which means nerves can be longer.
Myelin first appeared in the tree of life around the same time as jaws, and its importance in vertebrate evolution has long been recognized, but until now, it was unclear what molecular mechanisms triggered its appearance.
Tanay Ghosh from Altos Labs-Cambridge Institute of Science and colleagues noticed RetroMyelin’s role in myelin production when they were examining the gene networks utilized by oligodendrocytes, the cells that produce myelin in the central nervous system.
Specifically, they were investigating the role of noncoding regions including retrotransposons in these gene networks — something that hasn’t previously been explored in the context of myelin biology.
“Retrotransposons compose about 40% of our genomes, but nothing is known about how they might have helped animals acquire specific characteristics during evolution,” Dr. Ghosh said.
“Our motivation was to know how these molecules are helping evolutionary processes, specifically in the context of myelination.”
In rodents, the researchers found that the RNA transcript of RetroMyelin regulates the expression of myelin basic protein, one of…
Read the full article here