New research led by the the University of Wyoming, Laramie, provides additional evidence that tardigrade proteins eventually could be used to make life-saving treatments available to people where refrigeration is not possible and enhance storage of cell-based therapies.
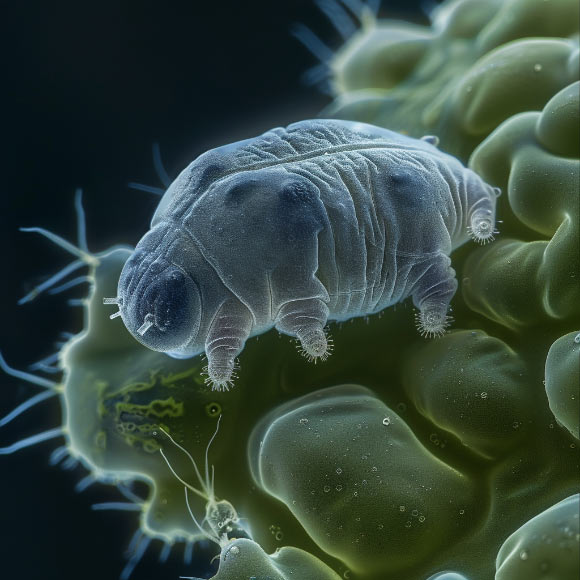
First discovered in 1773, tardigrades are a diverse group of microscopic invertebrates that are best known for their ability to survive extreme conditions.
Also known as water bears or moss piglets, these creatures can live for up to 60 years, and grow to a maximum size of 0.5 mm, best seen under a microscope.
They are able to survive for up to 30 years without food or water, for a few minutes at temperatures as low as minus 272 degrees Celsius (minus 457 degrees Fahrenheit) or as high as 150 degrees Celsius (302 degrees Fahrenheit), and minus 20 degrees Celsius (minus 4 degrees Fahrenheit) for decades.
Tardigrades withstand pressures from virtually 0 atm in space up to 1,200 atm at the bottom of the Marianas Trench, and are also resistant to radiation levels up to 5,000-6,200 Gy.
They survive by entering a state of suspended animation called biostasis, using proteins that form gels inside of cells and slow down life processes.
“Amazingly, when we introduce these proteins into cells, they gel and slow down metabolism, just like in tardigrades,” said Dr. Silvia Sanchez-Martinez, a researcher at the University of Wyoming, Laramie.
“Furthermore, just like tardigrades, when you put human cells that have these proteins into biostasis, they become more resistant to stresses, conferring some of the tardigrades’ abilities to the human cells.”
“Importantly, our research shows that the whole process is reversible,” said Dr. Thomas Boothby, a researcher at the University of Wyoming, Laramie.
“When the stress is relieved, the tardigrade gels dissolve, and the human cells return to their normal metabolism.”
“Our findings provide an avenue for pursuing technologies centered on…
Read the full article here