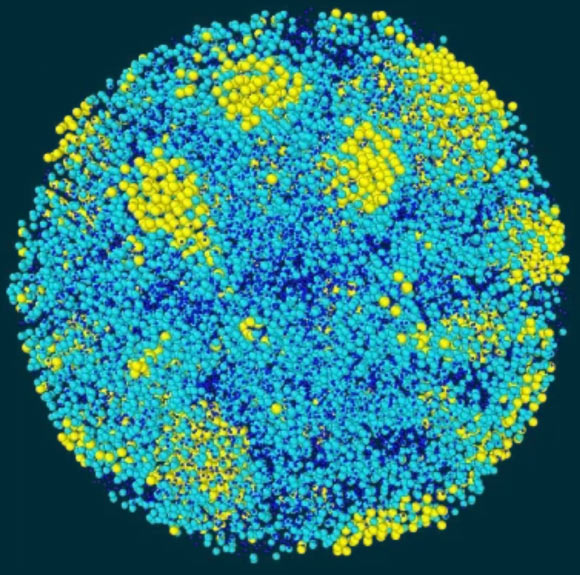
Ice 0 is a form of ice that can seed the formation of ice crystals in supercooled water, according to a team of researchers from the University of Tokyo.
“Crystallization of ice, known as ice nucleation, usually happens heterogeneously, or in other words, at a solid surface,” said University of Tokyo researchers Gang Sun and Hajime Tanaka.
“This is normally expected to happen at the surface of the water’s container, where liquid meets solid.”
“However, our research shows that ice crystallization can also occur just below the water’s surface, where it meets the air.”
“Here, the ice nucleates around small precursors with the same characteristic ring-shaped structure as ice 0.”
“Simulations have shown that a water droplet is more likely to crystallize near the free surface under isothermal conditions,” Dr. Sun added.
“This resolves a longstanding debate about whether crystallization occurs more readily on the surface or internally.”
The precursors of ice 0 have a structure very similar to supercooled water, allowing water molecules to crystallize more readily from it, without needing to directly form themselves into the structure of regular ice.
They are formed spontaneously, as a result of negative pressure effects caused by the surface tension of water.
Once crystallization begins from these precursors, structures similar to ice 0 quickly rearrange themselves into the more familiar ice I.
“The findings regarding the mechanism of surface crystallization of water are expected to contribute significantly to various fields, including climate studies and food sciences, where water crystallization plays a critical role,” Dr. Tanaka said.
“A more detailed understanding of ice and how it forms can give invaluable insight into a variety of areas of study.”
“This work may have particular importance in meteorology, for example, where ice formation via ice 0-like precursors may have a much more noticeable effect in small water…
Read the full article here