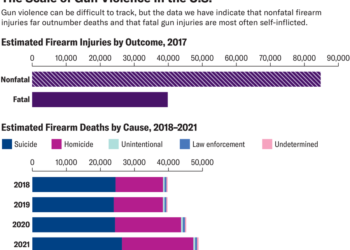
For the first time, researchers have harnessed the body’s own chemistry to “grow” electrodes inside the tissues of living fish, blurring the boundary between biology and machines.
The technique uses the body’s sugars to turn an injected gel into a flexible electrode without damaging tissues, experiments show. Zebrafish with these electrodes grown in their brains, hearts and tail fins showed no signs of ill effects, and ones tested in leeches successfully stimulated a nerve, researchers report in the Feb. 24 Science.
Science News headlines, in your inbox
Headlines and summaries of the latest Science News articles, delivered to your email inbox every Thursday.
Thank you for signing up!
There was a problem signing you up.
Someday, these electrodes could be useful for applications ranging from studying how biological systems work to improving human-machine interfaces. They also could be used in “bioelectronic medicine,” such as brain stimulation therapies for depression, Parkinson’s disease and other conditions (SN: 2/10/19).
Soft electronics aim to bridge the gap between soft, curvy biology and electronic hardware. But these electronics typically still must carry certain parts that can be prone to cracks and other issues, and inserting these devices inevitably causes damage to tissues.
“All the devices we have made, even though we have made them flexible, to make them more soft, when we introduce them, there will still be a scar. It’s like sticking a knife into the organ,” says Magnus Berggren, a materials scientist at Linköping University in Sweden. That scarring and inflammation can degrade electrode performance over time.
Previous efforts to grow soft electronics inside tissues have drawbacks. One approach uses electrical or chemical signals to power the transformation from chemical soup to conducting electrodes, but these zaps also cause damage. A 2020 study got around this…
Read the full article here