A widely used abortion medicine is in legal jeopardy, despite decades of data supporting its use.
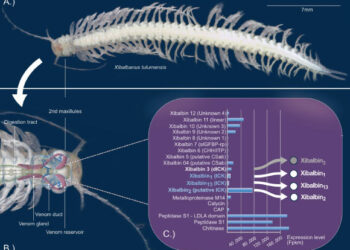
The medicine, called mifepristone, is part of two-drug regimen that can end a pregnancy, and was used in more than half of abortions in the United States in 2020. Late last year, a group of antiabortion doctors and others sued the U.S. Food and Drug Administration, challenging mifepristone’s initial approval in 2000 and the changes the agency later made to broaden access to the drug.
In the lawsuit, they claim that the drug isn’t safe, that the FDA improperly approved it and failed to follow the science, and that mifepristone is illegal to send in the mail. The U.S. District Court judge that first heard the case sided with the group. Now, the case is being reviewed by the U.S. Fifth Circuit Court of Appeals in New Orleans; on May 17, the Fifth Circuit heard oral arguments related to the judge’s ruling. The matter may ultimately be resolved by the Supreme Court.
But the original scientific evidence used in the FDA’s approval process offered a clear answer to the drug’s safety, says Thomas Kosasa, an obstetrician gynecologist at the University of Hawaii in Honolulu who served on a 1996 committee that advised the FDA. “We made sure that it was a safe medication,” he says. “It was also effective.”
In the 23 years since the agency approved mifepristone, the drug has amassed a long track record of safety data — in the United States and around the world, says Lauren Owens, an obstetrician gynecologist at the University of Washington Medical Center in Seattle. In 2020 in the United States alone, there were nearly 500,000 medication-induced abortions, which relied on mifepristone and a second drug, misoprostol, to terminate a pregnancy. The drugs are also used to manage miscarriages. And during the coronavirus pandemic, an influx of new safety data supported what earlier studies had found.
“Millions of people have used this…
Read the full article here