Importantly, the impaired neuronal responses are not restored after diet-induced weight loss, according to a new study led by Amsterdam University Medical Centers scientists.
Feeding behavior is a result of the integration of complex metabolic signals that travel from the gut, other organs and the circulation to the brain, and back, in order to trigger sensations of hunger and satiation and the motivation to look for food.
While these processes are starting to be better understood in animal models, including in the context of metabolic diseases such as obesity, much less is known about what happens in humans, given the difficulty in designing experimental setups in the clinic that could shed light on to these mechanisms.
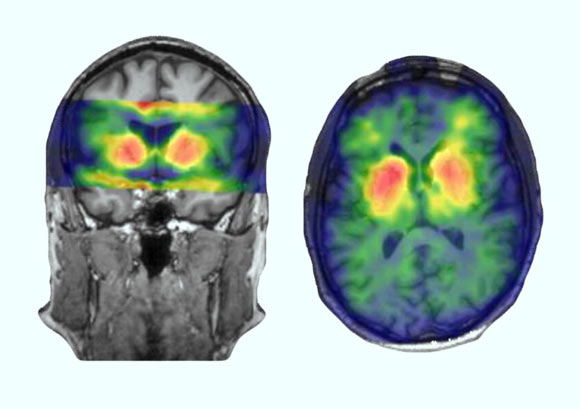
A research team headed by Dr. Mireille Serlie from Amsterdam University Medical Centers designed a controlled trial consisting of infusing specific nutrients (either lipids or carbohydrates) directly into the stomachs of 30 participants with a healthy bodyweight (defined as a BMI of 25 kg/m2 or below) or 30 individuals with obesity (a BMI of 30 kg/m2 or above), while simultaneously measuring their brain activity through functional MRI and SPECT.
While the participants with a healthy bodyweight displayed specific patterns of brain activity and dopamine release (associated with positive sensations) after nutrient infusion, these responses were blunted in participants with obesity.
Moreover, 10% body weight loss (following a 12-week diet) was not sufficient to restore these responses in individuals with obesity, suggesting long-lasting brain adaptations occur in the context of obesity and remain even after weight loss is achieved.
“We show that intragastric glucose and lipid infusions induce orosensory-independent and preference-independent, nutrient-specific cerebral neuronal activity and striatal dopamine release in lean participants,” the authors said.
“In contrast, participants with obesity have severely impaired brain responses to…
Read the full article here