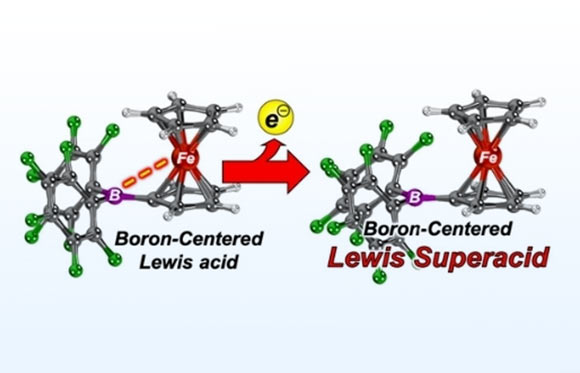
Lewis superacids are very special catalysts that can be used to break strong chemical bonds in non-biodegradable fluorinated hydrocarbons and climate-damaging greenhouse gases.
Lewis acids play a major role in all areas of chemistry.
For a long time, toxic, corrosive and oxidizing antimony pentafluoride (SbF5) was considered as the strongest Lewis acid known.
Lately, species significantly exceeding the Lewis acidity of antimony pentafluoride have been realized and were termed Lewis superacids.
“Lewis acids are compounds that add electron pairs,” said senior co-author Professor Jan Paradies and his colleagues from Paderborn University and the Karlsruhe Institute of Technology.
“Because of this ability, they are often used to speed up chemical reactions.”
These compounds are stronger than antimony pentafluoride and can break even the toughest bonds.
“For strong bonds, you need highly reactive reagents, i.e. substances that are extremely reactive,” Professor Paradies said.
“The new catalyst can split, for example, carbon-fluorine or sulfur-fluorine bonds, which are particularly robust.”
“Intrinsically, these Lewis superacids are incredibly reactive, which makes them difficult to produce and use.”
“Using a trick, we managed to produce such molecules and use them in catalytic reactions,” he added.
“This makes it possible to, for example, activate and further convert virtually inert, i.e. less reactive, carbon-fluorine or sulfur-fluorine bonds.”
The team’s work was published in the journal Angewandte Chemie.
_____
Laura Köring et al. Boron-Centered Lewis Superacid through Redox-Active Ligands: Application in C-F and S-F Bond Activation. Angewandte Chemie, published online January 9, 2023; doi: 10.1002/anie.202216959
Read the full article here