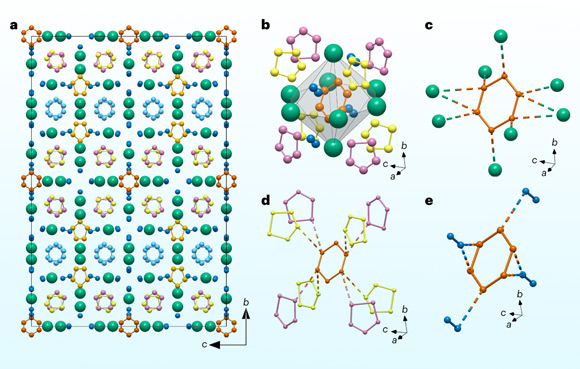
A team of chemists from Germany, Sweden, the United Kingdom and China has synthesized the potassium nitrogen compound K9N56 — composed of 520 atoms per unit cell — under high pressures (46 and 61 GPa) and high temperature (above 2,000 K).
“Although the term aromatic originally concerned odor, today its use in chemistry is restricted to compounds that have particular electronic, structural, or chemical properties,” said Professor Natalia Dubrovinskaia, a researcher at the University of Bayreuth and Linköping University, and colleagues.
“The unique stability of these compounds is referred to as aromaticity.”
“Aromatic compounds play an important role in chemistry and biology as well as in numerous branches of industry.”
“Although aromaticity was initially thought to be exclusive to carbon cycles, it has since been shown that numerous systems composed of carbon heterocycles and non-carbon cycles can have an aromatic character.”
“Nitrogen aromaticity, however, has far been restricted to the [N5]⁻ pentazolate anion.”
Professor Dubrovinskaia and her colleagues were able to synthesize the complex compound K9N56 — which contains [N6]4- hexazine rings — under high pressures (46 and 61 GPa) and high temperature (estimated to be above 2,000 K) in direct reaction between nitrogen and KN3 in a laser-heated diamond anvil cell.
K9N56 is composed of 520 atoms: 448 N and 72 K atoms, with the nitrogen atoms forming three distinct species: planar N6 rings, planar N5 rings and N2 dimers.
The structure of the [N6]4- hexazine ring conforms to the rule for aromaticity of chemical compounds named after physical chemist Erich Hückel: the ring is cyclic, planar and has (4n + 2) π-electrons (10π system).
“This is the first time that a ring consisting of six nitrogen atoms has been synthesized that follows the Hückel rule for aromaticity,” Professor Dubrovinskaia said.
“Its aromatic character is further supported by bond-length…
Read the full article here